The ocean is one of the most vital components of the Earth’s ecosystem. It produces more than half of the planet’s oxygen, absorbs excess heat, maintains global temperatures, and provides food and livelihood to billions of people. However, over the last few decades, the ocean has been experiencing a rapid and dangerous environmental change — Ocean Acidification. Unlike storms or tsunamis, it cannot be seen directly, but its effects are far more widespread and long-lasting.
Ocean acidification is strongly linked to climate change, and together they pose a serious challenge for the sustainability of life on Earth. Understanding this problem is the first step toward saving our oceans and the many lives that depend on them.
What Is Ocean Acidification?
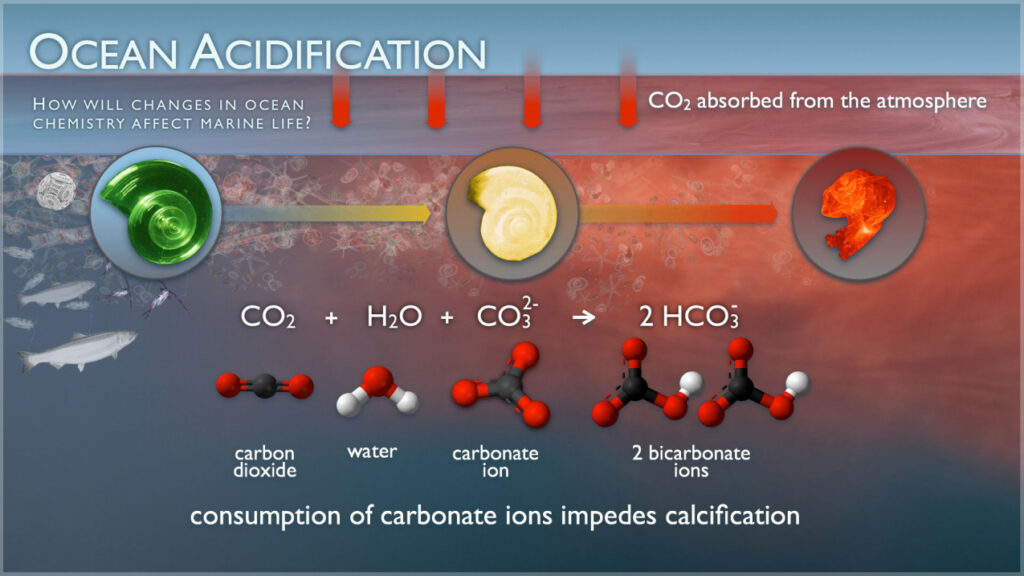
Ocean acidification refers to the decrease in the pH level of seawater caused by the absorption of high amounts of carbon dioxide (CO₂) from the atmosphere. When CO₂ dissolves in seawater, it undergoes a chemical reaction and forms carbonic acid, which reduces the ocean’s pH and increases acidity.
Oceans currently absorb 30% to 40% of the total CO₂ emitted into the atmosphere every year.
While this helps reduce the greenhouse gases in the atmosphere, it puts the ocean under extreme chemical stress.
Ocean water has naturally been slightly alkaline for millions of years, providing a stable environment for marine organisms. However, due to rapid industrialization and fossil fuel burning, CO₂ levels have increased dramatically, disturbing this natural balance. The ocean has already become 30% more acidic than the pre-industrial period — and the rate is increasing every year.
Main Causes of Ocean Acidification
Ocean acidification is mostly caused by human activities that release carbon dioxide into the atmosphere. The leading causes include:
1️⃣ Burning of Fossil Fuels
Industries, transportation vehicles, power plants, and factories burn coal, oil, and natural gas, releasing billions of tonnes of CO₂ into the air. Much of this CO₂ later dissolves in ocean water.
2️⃣ Deforestation and Forest Degradation
Plants and trees naturally absorb CO₂. Large-scale cutting of forests reduces this natural absorption process, leaving more CO₂ in the atmosphere — eventually reaching the oceans.
3️⃣ Industrial and Manufacturing Growth
Since the Industrial Revolution, human emissions have increased exponentially. Today’s rapid industrial expansion continues to worsen ocean acidity.
4️⃣ Agriculture and Chemical Fertilizers
Agricultural practices release nitrous oxide and CO₂ into the atmosphere, indirectly contributing to acidification.
5️⃣ Cement and Construction Industries
Cement production is one of the largest industrial sources of CO₂ emissions globally.
All these factors together are pushing ocean chemistry far beyond its natural limits.
How Ocean Acidification Affects Marine Life
Marine life has evolved to survive in a specific pH range. When this pH changes, even slightly, biological functions and survival processes are affected.
Many organisms rely on calcium carbonate to build shells, bones, and skeletons. Acidic water dissolves this essential mineral, making it incredibly difficult for them to grow and survive.
Species severely affected:
- Corals
- Oysters, clams, mussels
- Snails and mollusks
- Sea urchins
- Lobsters and crabs
- Plankton (tiny organisms forming the base of the marine food chain)
When calcium carbonate becomes scarce:
- Shells become weaker and thinner
- Growth slows down dramatically
- Reproduction becomes difficult
- Mortality rate increases
According to marine scientists, if ocean acidity continues at the same rate, coral reefs may stop growing by the end of this century.
Impact on Marine Food Chains and Biodiversity
Once the smallest organisms like plankton and shell-forming species start dying, the entire food chain collapses. Big fishes, seabirds, and marine mammals depend on thesThe collapse of small and microscopic organisms has a ripple effect throughout the ocean. Plankton, shell-forming species, and coral ecosystems support thousands of larger animals.
Consequences of ocean acidification include:
- Loss of habitat for marine species
- Decline in fish populations
- Disturbance in the reproduction of marine species
- Disruption of predator–prey relationships
- Extinction of sensitive species
Biodiversity loss ultimately leads to a weak and unstable ecosystem, unable to support future generations of marine life.
Impact on Humans and Global Economy
More than 1 billion people depend on seafood as their primary source of protein. Millions of people rely on fishing for employment. Ocean acidification puts all of this at Ocean acidification is not just an environmental issue — it is an economic and social crisis.
More than 1 billion people depend on seafood as their main source of protein, especially in coastal regions. If fish and shellfish populations decline, food insecurity will rise sharply.
Economic consequences:
- Collapse of commercial fisheries and aquaculture industries
- Job losses in fishing, seafood processing, and coastal tourism
- Increase in seafood prices worldwide
- Loss of income in coral-dependent tourism destinations (e.g., Maldives, Great Barrier Reef, Caribbean)
Experts estimate that global fisheries could lose billions of dollars annually due to ocean acidification.
Ocean Acidification & Climate Change: A Dangerous Connection
Ocean acidification doesn’t just affect marine life — it also affects the global climate system. Normally, oceans act as carbon sinks, helping reduce CO₂ in the Oceans act as a natural climate regulator by absorbing CO₂ and heat from the atmosphere. But when the ocean becomes too acidic:
- It loses its ability to absorb CO₂ efficiently
- More CO₂ remains in the atmosphere
- The greenhouse effect becomes stronger
- Global temperatures rise even faster
This creates a harmful feedback loop, accelerating:
- Global warming
- Melting of polar ice
- Sea level rise
- Extreme weather disasters
Thus, protecting oceans is essential not just for marine life but also for controlling climate change.
How Can We Reduce Ocean Acidification?
Ocean acidification is one of the most urgent environmental challenges of our time, but the good news is that it can still be slowed down and controlled. However, this requires collective action at the global, national, industrial, and individual levels. Protecting the ocean means protecting the stability of the entire planet — climate, biodiversity, food security, and economic livelihood. The solutions below explain how humans can reverse the harmful impact of ocean acidification.
1. Reduce Carbon Emissions — The Most Crucial Step
The primary cause of ocean acidification is CO₂ emissions from human activities. The most effective solution is to reduce global carbon output. This can be achieved by shifting from fossil fuels to renewable energy like solar, wind, geothermal, and hydropower. Improving energy efficiency in homes, industries, and transportation, adopting electric vehicles, and promoting carbon-capture technologies in factories can significantly slow the acidification process. The less carbon released into the atmosphere, the less the ocean absorbs.
2. Protect and Restore Forests
Forests act as natural carbon sinks and play a vital role in lowering atmospheric CO₂. Stopping deforestation, increasing afforestation, and restoring degraded forests—especially mangroves—can greatly reduce the amount of CO₂ dissolving into ocean waters. Strong forest protection laws and community-based conservation programs make this effort more effective.
3. Promote Sustainable Fishing
Balanced marine ecosystems are better equipped to resist the impacts of acidification. Enforcing scientific fishing limits, preventing illegal fishing, using non-destructive gear, and protecting nursery zones help maintain marine biodiversity and ecosystem resilience.
4. Reduce Ocean Pollution
Plastic waste and chemicals weaken marine organisms already stressed by acidification. Minimizing single-use plastics, improving waste management, regulating industrial discharge, and supporting coastal cleanup programs can restore water quality and protect sea life.
5. Encourage Green Industries
Industries must shift to eco-friendly production and carbon-neutral technologies. Governments can support sustainable business practices through incentives, emission regulations, and long-term climate goals that promote low-carbon economic growth.
6. Increase Awareness and Conservation Efforts
Widespread public awareness drives real change. Education programs, documentaries, sustainable seafood choices, and international cooperation can accelerate action. Investments in marine protected areas, coral restoration, and ocean monitoring systems are essential to strengthen marine life and safeguard the oceans for the future.
Conclusion
Ocean acidification is one of the most urgent and critical environmental threats facing our planet today. It is a direct result of excessive carbon emissions from human activities and is steadily damaging marine life, disturbing biodiversity, harming coastal economies, and accelerating the climate crisis.
If this trend continues, the world could face:
- A breakdown of marine ecosystems
- Severe decline in seafood resources
- Collapse of ocean-based industries
- Faster global warming and climate instability
But there is still time to act. By reducing carbon emissions, protecting forests, conserving marine life, and supporting sustainable practices, we can restore the balance of ocean chemistry and protect life on Earth.